Methane Gas Detection – Know The Risks & Detect The Danger
Published 26 Sep 2016

T&D: Gas Detection Specialists
- By Chris Dodds : estimated reading time 5 minutes
Methane Gas Detection
The following article has been written for anyone working in industries where exposure to methane gas is a risk. The article begins by identifying common industries where methane gas is present or used and goes on to discuss the hazards it poses to plant and personnel.
Finally, the article looks at Crowcon gas detection technologies and solutions including infrared and Pellistor type gas sensors.
The largest ever marine oil spill occurred on the night of April 20th, 2010 when a methane gas release triggered a deadly explosion on the BP Macondo site – for 87 days oil spewed over 68,000 square miles of ocean and 16,000 miles of coastline off the Gulf of Mexico.
Disaster By Numbers
- 11 – platform workers killed, bodies never found
- 36 – hours the oil platform burned before sinking
- 4, 900,000 – total barrels of crude oil released before the leak was capped on 15 July
- 53,000 – leaked barrels per day, BP initial prediction was 1,000 barrels
- 3,850 – square mile spread of leaked oil along 125 mile coastline by 4 June
- 4,768 – dead animals collected as of 13 August
Disaster In Dollars
- $2.5 billion – initial estimated cost of the oil spill to the US fishing industry
- $23 billion – estimated cost to local tourist industry (US Travel Association)
- $105 billion – total share-price value lost by BP between April and June
- $69 billion – bill sent to BP by the Obama administration for the clean up effort
Read BP’s Deepwater Horizon Accident & Response Statement.
In the correct concentration, methane gas can be very dangerous and can cause huge explosions if ignited. It has been the cause of many disasters in the mining, water, oil and gas industries.
In 1984, 16 people were killed in the Abbeystead disaster (8 at the scene and 8 later of injuries) – here methane gas had seeped into the valve house of a pumping station from underground coal beds and became ignited.
T&D working in partnership with Crowcon aim to provide useful and informative articles about the detection of a range of toxic, flammable and explosives gases – as well as methane gas detection we discuss the risks posed by hydrogen sulphide, carbon monoxide, ammonia and oxygen depletion.
Electrical, Heating & Process Instrumentation Specialists
Thorne & Derrick are stockists and suppliers of an extensive range of equipment for use in industrial, process and hazardous area industries where potentially explosive atmospheres necessitate the use of products certified for safe use in the presence of flammable gases and vapours according to hazardous area classifications such as ATEX or IUECEx.
What Is Methane Gas?

Methane Gas CH4
Methane is a colourless, tasteless, odourless gas and has the chemical formula CH4 – methane is the main component of natural gas. For those who didn’t listen to their chemistry teacher at school, it is made up of one atom of carbon and four atoms of hydrogen.
Methane is produced naturally by the process of methanogenesis and is found under the ground and in seabed locations – it is commonly used in chemical industries and also for electricity generation.
Methane is non-toxic but highly explosive (more on that later.)
Production of methane gas occurs naturally in many industries including waste disposal, mining, oil and gas, petrochemical and the energy sector.
Detection, Analysis & Monitoring Of Landfill Methane Gas
Despite the established trend towards household waste recycling, DEFRA (Department of The Environment, Food & Rural Affairs) reported that 8.7 million tonnes of Biodegradable Municipal Waste (BMW) were sent to landfill in 2014.
Methane is both a serious potential explosion risk and as a landfill gas should be effectively monitored and analysed using either a portable or permanent gas detection system in accordance with UK government emission targets.
3 Gas Monitoring Options
- Borehole Monitoring – methane gas concentration levels can be monitored in the borehole headspace, the relative concentration of carbon dioxide, oxygen and methane indicates the extent of the decomposition process. On site methane gas emissions can be detected using handheld portable detectors or fixed detection systems which provide gas readings for logging and analysis.
- Flux-Box Monitoring – here methane gas is detected through breaches in the cap of a closed landfill therefore quantifying the total emission extent from the surveyed area. Landfill gas methane emissions are monitored using either static closed chamber or dynamic closed chamber flux-boxes – cap defects and specific point gas emissions monitored by flux-boxes provide reliable methane gas monitoring and analysis for low landfill gas fluxes.
- Perimeter Monitoring – the landfill site is monitored for methane gas around the perimeter of the landfill area – the permitted limit for ambient methane gas leaks and escapes from a landfill site is 10ppm.
Where is Methane Gas Used?
Methane gas is commonly used in chemical industries and is used to refine petrochemicals. It is also used as a fuel and is burned in gas turbines or steam generators to produce electricity.
Methane is widely used domestically for heating and cooking in homes (but you knew that didn’t you).
Methane is the main component of Liquefied Natural Gas (LNG) and Compressed Natural Gas (CNG). Methane is generated by the decomposition of biodegradable solid waste as well as animal and human waste. It is therefore commonly present in landfill sites and sewage treatment works.
As methane is an incredibly potent, hazardous greenhouse gas it is important to not only manage storage and production but also re-use and recycle the gas. Methane emissions represent a profitable solution to global climate change.
The most common anthropogenic sources of methane gas are agriculture, mining, landfills and natural gas oil activities.
Why Is Methane Gas Dangerous?
Methane is not generally considered a toxic gas, however it is extremely flammable even in low concentrations when mixed with other chemicals – it is also an asphyxiant as it will displace oxygen. This is particularly dangerous in confined spaces working.
In order to create a fire/explosion, you need three things: 1. oxygen 2. an ignition source and 3. a fuel.
Take away the oxygen and you remove the risk of explosion – in contrast high levels of oxygen will cause fuels to burn faster and more vigorously. For an explosive atmosphere to exist, a certain ratio of oxygen and fuel must exist. The ratio differs depending on the fuel.
In the gas detection industry, such ratios are known as lower explosion limits (LEL) and upper explosion limits. (UEL)
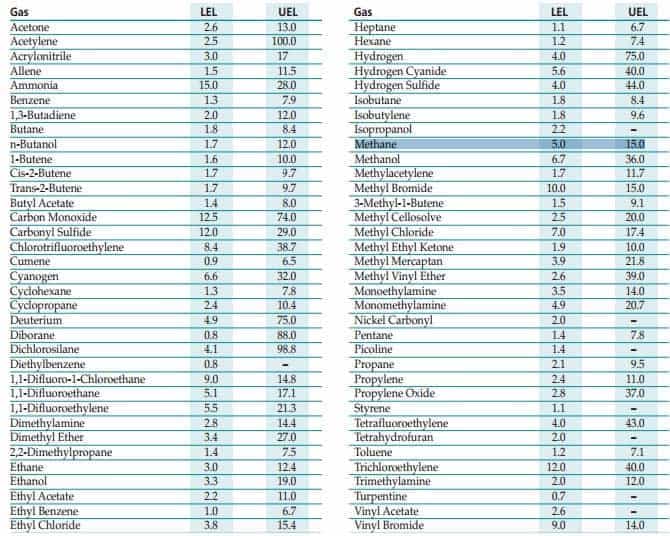
Methane Gas: LEL & UEL
LEL is defined as “the minimum concentration of a particular combustible gas necessary to support its combustion in air.” Concentrations below this level will not burn. The UEL is defined as “the highest concentration (percentage) of a gas or a vapor in air capable of producing a flash of fire in presence of an ignition source.”
The range between LEL and UEL is referred to as the flammable range and as the name suggests is when fire/explosions will occur.
As can be seen from the table, the LEL for methane is 5% and UEL is 15%. Concentrations of 9% are thought to be the most volatile. It may sound strange but concentrations above 15% will not be explosive as the air is too saturated with methane. However this is when asphyxiation can be just as hazardous.
Asphyxiation becomes a risk when there are high concentrations of methane. This is because the methane displaces the oxygen. We need approximately 18% oxygen to breath, levels below 16% can be dangerous and levels below 10% can cause immediate loss of consciousness and inevitably death. Working in confined spaces can be extremely dangerous if exposure to methane (or any other gas for that matter) is considered a risk.
Coal Mining & Methane
Coalbed methane occurs naturally in coal seams. Methane recovered from underground coal mines is generally grouped under the term Coal Mine Methane (CMM). 2 key factors influence CMM recovery : mine safety and the opportunity to mitigate significant volumes of methane emissions arising from coal mining activities.
Methane emissions in mines arise at 2 two key stages :
(1) Methane is released as a direct result of the physical process of coal extraction. In many modern underground mines, the coal is extracted through longwall mining. Longwall mining, as with other sub-surface techniques, releases methane previously trapped within the coal seam into the air supply of the mine as layers of the coal face are removed, thus creating a potential safety hazard.
(2) Methane emissions arise from the collapse of the surrounding rock strata after a section of the coal seam has been mined and the artificial roof and wall supports are removed as mining progresses to another section. The debris resulting from the collapse is known as gob and also releases methane or ‘gob gas’ into the mine.
32 miners unaccounted for, 1 dead after coal mine explosion in eastern #Ukraine. Methane gas is suspected cause: http://t.co/g3lTgxT6Dl
— CBC News Alerts (@CBCAlerts) March 4, 2015
Advice & Considerations For effective Methane Gas Detection
There are no specific guidelines for detection of methane but the UK Health & Safety Executive (HSE) does provide information for the selection and use of flammable gas detectors. The HSE provides advice and information on the selection, installation, use and maintenance of industrial flammable gas detectors – it is aimed at process engineers and managers and others concerned with the use of flammable gas detectors.
Fixed and portable methane gas detectors should be used to help minimise risk and provide early warnings should gas levels become dangerous. They can be a life saving pieces of equipment and it is important that the correct gas detection measures are implemented to ensure your plant is protected but more importantly that people return home from work.

Thorne & Derrick are distributors for Crowcon Detection Instruments Ltd – specialists in the manufacture of flammable and toxic gas detection equipment.
Part of this measure should also include adequate training for the users. As some people pointed out following a recent blog article about portable gas detectors, “the gas detector will not prevent accidents if the user doesn’t know how to use it”.
This may sound obvious but there are many stories about people incorrectly using gas detectors, ignoring warning alarms, failing to bump test and calibrate sensors.
Gas detectors usually measure in either % volume or PPM (parts per million).
Flammable gases are usually measured by % volume and toxic gases by PPM. As mentioned earlier the LEL for Methane is 5%. Typically warning levels on gas detectors can be set between 0-100% of the LEL.
The HSE recommend that first alarm level should be set no higher than 10% of the LEL and the second alarm level should be no more than 25% of the LEL.
There are two main types of detector technology used for measuring flammable gases : Infrared and Pellistor.
Infrared Gas Detectors
Gases such as methane which contain more than one type of atom can be detected by IR gas sensors. This is because the gas will absorb infrared radiation. An infrared gas detector such as the Crowcon IRmax are made up of an infrared source (transmitter) and an infrared detector (receiver).
If methane passes between the transmitter and receiver, it absorbs the radiation and the intensity of the signal at the receiver is weakened. Specific gases are detected by measuring the amount of absorbed infrared radiation at specific wavelengths, the difference being related to the concentration of gas present.
There are many benefits to using infrared gas detection technology and advancements in technology mean that they are no longer as complicated, cumbersome or expensive and gases detected with infrared equipment are typically corrosive and reactive.
The main advantage of IR gas detectors is that the equipment does not directly interfere with gas that is being detected. The major functional components of the analyser are protected with optical lights and therefore gas molecules only ever interact with a light beam.
Pellistor Detectors
Pellistor sensors are commonly used in both fixed and portable gas detectors such as the Crowcon Flamegard Plus, Crowcon Clip, Gasman and Tetra 3.
Pellistors can be used to detect combustible gases such as Methane.
The principal of operation is based around changes in resistance caused by target gases on the small pellets of catalyst loaded ceramic. As the gas comes into contact with the sensor, it is burned which generates heat and alters the resistance of the detecting element of the sensor which is proportional to the target gas.
Pellistor sensors are accurate and remain unaffected by changes to ambient temperatures, humidity or pressure. The main drawback to pellistor technology is the possibility of contamination or poisoning – pellistor gas detectors are susceptible to sulphides, silicones, hydrocarbons and lead.
Therefore a routine maintenance programme including regular calibration should be considered when using this type of technology. This can and will add to the lifetime cost of the product. A further disadvantage is that they will fail to work properly if oxygen isn’t present and therefore they are not always suitable for gas detection in confined spaces. In contrast Infrared sensors do not require the presence of oxygen and should therefore be used when oxygen depletion is a possibility.
IR sensors also have a failsafe function whereby if the detector becomes obscured or fails, no radiation will register and an alarm will be raised.
The following Crowcon video is an illustration of how the pellistor, based on a Wheatstone bridge circuit, includes two “beads”, both of which encase platinum coils. One of the beads (the ‘active’ bead) is treated with a catalyst, which lowers the temperature at which the gas around it ignites.
This bead becomes hot from the combustion, resulting in a temperature difference between this active and the other ‘reference’ bead. This causes a difference in resistance, which is measured; the amount of gas present is directly proportional to it, so gas concentration as a percentage of its lower explosive limit (%LEL) can be accurately determined.
- Pellistor sensor operation from Crowcon Detection Instruments on Vimeo.
Pellistor sensors are accurate are remain unaffected by changes to ambient temperatures, humidity or pressure. The main drawback to pellistor technology is the possibility of contamination or poisoning.
They are susceptible to sulphides, silicones, hydrocarbons and lead. Therefore a routine maintenance programme including regular calibration should be considered when using this type of technology.
This can and will add to the lifetime cost of the gas detection process.
A further disadvantage is that they will fail to work properly if oxygen isn’t present and therefore they are not always suitable for gas detection in confined spaces. In contrast Infrared sensors do not require the presence of oxygen and should therefore be used when oxygen depletion is a possibility.
IR sensors also have a failsafe function whereby if the detector becomes obscured or fails, no radiation will register and an alarm will be raised.
Further Reading & Guidelines
BS EN 50073: 1999 Guide for the selection, installation, use and maintenance of apparatus for the detection and measurement of combustible gases or oxygen.
BS EN 50241: 1999 Specification for open path apparatus for the detection of combustible or toxic gases and vapours.

The EXHEAT Bulldog – hazardous area electrical heating for Zone 1 & Zone 2 potentially explosive atmospheres
The Crowcon Gas Academy
Interested to learn more about gas detection? Are you involved with the specification or design of gas detection systems for your plant? Crowcon provide training courses tailored to suit your needs. Crowcon manufacture single gas and multigas detectors for personal monitoring and portable safety applications providing protection against a wide range of industrial gas hazards.

The Crowcon Gas Academy – training courses cover the full Crowcon fixed systems and portable gas detection product ranges
Know-How & Expertise
T&D take customer support and service seriously and are committed to excelling their product knowledge to ensure the safe and optimum sale of gas detectors for all applications – all T&D Sales Engineers are certificated by Crowcon for Selling Crowcon Products.

T&D: Trained By Crowcon, The Gas Detection Specialists

Read: Detecting Gas – Hydrogen Sulphide, Methane, Oxygen & Carbon Monoxide Gas Using Crowcon Detectors
Flame, Heat, Gas Detectors & Detection Systems
![]()
Hazardous area industries including offshore oil/gas platforms and FPSOs, onshore oil refineries, processing plants, pipelines, storage farms and LPG/LNG plants all utilise or produce a wide range of hazardous flammable liquids and gases that can be detected using correctly specified flame and gas detectors.
Detecting toxic and flammable gases requires the detectors to be classified and certified according to the specific IECEx or ATEX standard – we distribute flame and fire detectors manufactured by Spectrex to operate in the harshest environmental conditions including self-contained stand-alone devices designed for direct connection to control and alarm systems or automatic fire extinguishing systems.
Our range of Hazardous Area Fire & Gas Detection System products also includes Explosion Proof Warning Systems & Hazardous Area Lighting – comprehensive range of intrinsically safe, flameproof and explosion proof alarm sounders, sirens, bells and horns, loudspeakers and beacons.
♦ LV Power Products | Junction Boxes & Enclosures ATEX | Plugs ATEX | Control Stations ATEX | Isolators ATEX

Thorne & Derrick | Detecting Flame | Heat | Gas
Experts in Equipment
for Explosive Atmospheres
FOLLOW US
Follow our Showcase Page on LinkedIn to receive hazardous area product innovations, industry news, whitepapers, videos, technical tips and training webinars for professionals involved in the explosive atmosphere industries.








